No needles. No cuffs. Just health tracking that blends into daily life
World’s first non-invasive device and platform that
measures key health markers, including blood glucose
and blood pressure.




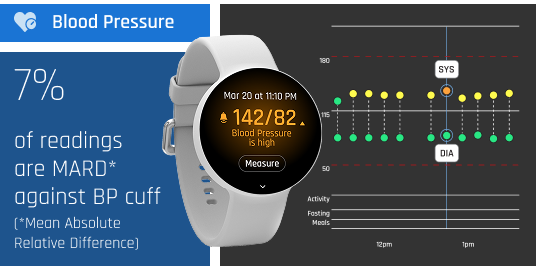








Less barriers and improved adherence for a healthier population
Remote monitoring devices can feel awkward and difficult to incorporate into everyday life, making patient adherence challenging.
Without consistent use, it’s hard to find trends and fight chronic diseases. LifeLeaf is a clinically proven, globally accessible and low-cost medical device with the look and feel of an elegant smartwatch.
LifeLeaf easily tracks patients’ health with the comfort of a lightweight and discreet device. It’s comfortable enough to wear around the clock, making patient adherence easy. With simple and painless adherence comes continuous data, observable trends, and ultimately, healthier populations.


A better way to
manage chronic care
Fully integrated remote patient
monitoring platform
While other home-use medical devices measure data in discrete points, LifeLeaf gathers continuous, high-volume medical-grade data, right from the wrist, making it easy to detect trends.
From fitness to nutrition and overall lifestyle, the LifeLeaf platform easily connects with 3rd party data sources to deliver a bigger picture.

THE APP
Provides simple visuals explaining trends, actionable insights, and the ability to share health records.

THE CLOUD
Patient health trends, accessible from anywhere.


USER PORTAL
Patients can view long-term trends and detailed health records.

THE DEVICE
Single non-invasive, effortless wrist-worn device that monitors multiple key biomarkers.

CAREGIVER PORTAL
Allows care teams to view real-time health trends and engage in proactive care management.

LifeLeaf provides simple visuals explaining trends, actionable insights and alerts, and the ability to share health records from anywhere.
Single non-invasive, effortless wrist-worn device that monitors multiple key biomarkers.

Patients can view long-term trends and detailed health records from anywhere.

Allows care teams to view real-time health trends and engage in proactive care management from anywhere.
Built on a strong foundation of
clinical evidence
LifePlus has completed multiple clinical feasibility studies in the US and across the world with over 70,000 paired data points from 500+ subjects in both clinical and real-world settings.
LifeLeaf’s impressive performance and extraordinary method of measuring biomarkers has also been highlighted in the world’s leading clinical meetings.
As LifeLeaf continues to provide people with better health tracking, additional global multi-site studies are enrolling to demonstrate the improved outcomes LifeLeaf delivers.
LifePlus has completed multiple clinical feasibility studies in the US and across the world with over 70,000 paired data points from 500+ subjects in both clinical and real-world settings.
LifeLeaf’s impressive performance and extraordinary method of measuring biomarkers has also been highlighted in the world’s leading clinical meetings.
As LifeLeaf continues to provide people with better health tracking, additional global multi-site studies are enrolling to demonstrate the improved outcomes LifeLeaf delivers.

T. BAILEY, J. FISCHER, I. SEN-GUPTA, B. MBOUOMBOUO, A. SANYAL
DIABETES, VOL. 74, SUPP. 1, 2025
Using real-world data, demonstrated feasibility of a truly non-invasive CGM on a diverse population from 248 end users from 12 countries.

T. BAILEY, J. FISCHER, M. KOHN, I. SEN-GUPTA, J. BROZ, A. SANYAL, B. MBOUOMBOUO, N. BANERJEE
DIABETES TECHNOLOGY & THERAPEUTICS, VOL. 25, SUPP. 2, 2023, PP. A-250
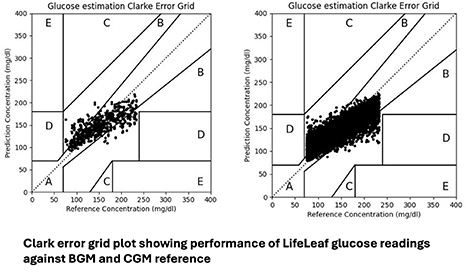
IRB-controlled multi-site feasibility study demonstrated clinical grade performance of LifeLeaf blood glucose readings against BGM and CGM reference.
W. ALMAHMEED, D. KUMAR, B. MBOUOMBOUO, N. BANERJEE, A. SANYAL, I. SEN-GUPTA
JOURNAL OF HYPERTENSION, VOL. 41, SUPP. 3, 2023, PP. E111-112
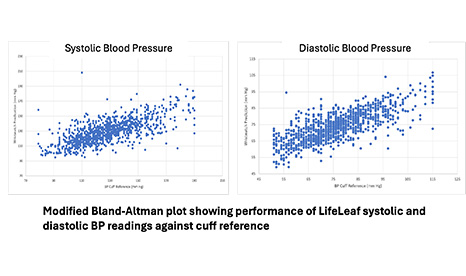
IRB-controlled multi-site feasibility study demonstrated clinical grade performance of LifeLeaf blood pressure readings against cuff reference.
Paradigm shift in chronic care management

With LifeLeaf, there’s no need to manage an ongoing supply chain of finger sticks, needles or sensors. It’s a single piece of hardware. That’s it.

Delivers a seamless capture, retention and presentation of health data to ensure maximum reimbursement.

Less time spent managing adherence means more time for more patients. LifeLeaf allows care teams to be more efficient and effective.
Your details have been submitted successfully!